Post-Partum Hyponatraemia in the Setting of Massive Haemorrhage: An Unusual Presentation of SheehanÃÆâÃâââ¬Ãâââ¢s Syndrome
Ronit L Cukierman, Kym T. Jones, M. Epee-Bekima and Timothy Phillips
Ronit L Cukierman*, Kym T. Jones, M. Epee-Bekima and Timothy Phillips
Department of Gynecology & Obstetrics, King Edward Memorial Hospital, Perth, Western Australia
- Corresponding Author:
- Ronit Cukierman
Department of Gynecology & Obstetrics
King Edward Memorial Hospital, Perth, Western Australia,
E-mail: ronitcukierman@gmail.com
Received Date: February 16, 2016; Accepted Date: February 26, 2016; Published Date: March 02, 2016
Citation: Cukierman RL, Jones KT, Bekima ME, et al. Post-Partum Hyponatraemia in the Setting of Massive Haemorrhage: An Unusual Presentation of Sheehan’s Syndrome. Crit Care Obst&Gyne. 2016, 2:1. doi:10.21767/2471-9803.100015
Abstract
A 30yr old woman, MK, complained of nausea day 8 post massive post-partum haemorrhage (PPH) on a background of elective section for suspected placenta accreta. Despite being clinically stable and prepared for discharge, she was found to be profoundly hyponatraemic on a baseline laboratory screen. MK was transferred to a high acuity unit and a diagnosis of Sheehan’s syndrome was later confirmed. Although clinical presentation of hypopituitarism may be subtle, life-threatening hyponatraemia or ACTH-insufficiency may have catastrophic outcomes if not considered as a potential diagnosis.
Keywords
Post-partum; Sheehan’s syndrome; Hypopituitarism; Hemorrhage
Background
Sheehan syndrome (post-partum hypopituitarism) is a rare complication of massive haemorrhage, occurring secondary to infarction of the enlarged pituitary of pregnancy. It is a result of profound hypovolaemic hypotension. The damage may be mild to severe and affect the secretion of one or more hormones [1].
Anatomy
The pituitary gland, comprised of the anterior and posterior lobes is separated by the pars intermedia and supplied by the pituitary stalk. This 10 mm organ resides in the sella turcica, a saddle-like depression in the skull base.
The anterior pituitary (adenohypophysis), derived from an embryonic invagination of pharyngeal epithelium (Rathke’s Pouch) [2] is responsible for the secretion of:
1. Growth hormone (GH)
2. Adrenocorticotrophin (ACTH), affecting the secretion of some glucocorticoids
3. Thyroid-stimulating hormone (TSH)
4. Prolactin
5. Gonadotropins (FSH, LH)
The posterior pituitary (neurohypophysis) originating from the neural tissue outgrowth from the hypothalamus is responsible for the production of both vasopressin (antidiuretic hormone) and oxytocin.
The most common post-partum presentation with Sheehan’s is a failure to lactate [3] accompanied by a variation of symptoms (Table 1) depending on the hormones affected, including: lethargy, anorexia, weight loss and/or secondary amenorrhea, within days or weeks after delivery [4]. Mild damage to the pituitary can explain the delay in symptoms [5].
| Corticotrophins: Mild: Severe: Postural Hypotension Tachycardia Fatigue Vascular Weakness Collapse Hypoglycaemia Death Dizziness |
Gonadotropins Amenorrhea Oligomenorrhoea Hot flushes Decreased libido Decrease genital/axillary hair |
| Growth Hormones: Fatigue Decreased quality of life Decreased muscle mass |
TSH: Fatigue Decreased Appetite Constipation Dry skin Bradycardia |
Table 1: Signs and symptoms of anterior hypopituitarism.
Studies have reported a large variation in the time from the suspected pituitary insult to the diagnosis of Sheehan’s, ranging from 1-33 years [6] and 2-40 years [4]. A review conducted by Huang of 14 patients with postpartum hemorrhage demonstrated severe hyponatremia (serum sodium <125 mmol/l) more than 16 years after the primary event in 8 of the 14 patients [6]. These numbers were not reflected in the review by Sanyal and Raychaudhur [7], who after examining 18 cases of Sheehan’s syndrome found that only four (22.22%) presented to an emergency department with hyponatremia (all of these patients were over 60 years of age at presentation), however 17 presented with lactational failure and 13 did not menstruate following their last delivery. The study of 20 patients conducted by Dökmetas et al., demonstrated hyponatremia in 35% of the cases studied, where as 70% of the patients lacked post-partum milk production [4]. Both studies showed a lack of menstruation following delivery. Sanyal and Raychaudhur found that 5 of 18 patients had a period of post-partum amenorrhea, on average, for a minimum of 3 years.
Although patients most commonly present with failure to lactate or amenorrhea, hyponatremia is the most common electrolyte disturbance [5]. Given the less specific symptoms and signs of hyponatremia, clinicians must be cognisant of the broad clinical spectrum that can signify a diagnosis of Sheehan’s syndrome.
Case
MK, 30 year old G2P2 healthy woman, underwent an elective lower uterine caesarean section for suspected placenta accreta. Her delivery was coordinated through a multi-disciplinary approach with input from haematology, anaesthetics and senior obstetric staff with meticulous planning. At section, a vigorous live infant was delivered with Apgar scores of 9 and 9 at 1 and 5 minutes, respectively. MK’s elective section was complicated by an initial PPH of 3000mls requiring a Bakri balloon and left uterine haemostatic sutures.
MK became profoundly hypotensive in recovery with ongoing vaginal loss and urgently returned to theatre. The Bakri balloon was removed with an immediate loss of approximately 800ml of blood accompanied by ongoing poor uterine tone. The decision was made to proceed to total abdominal hysterectomy and bilateral salpingectomy.
Intraoperatively, MK remained hypotensive and coagulopathic, necessitating our massive transfusion protocol including blood, platelets, and fresh frozen plasma. The additional estimated blood volume was 2.5L, totaling 5.5L. Her systolic blood pressure dropped from 80mmHg to the lowest documented reading of 50mmHg an hour intraoperatively before returning to a normal range. Her lowest documented haemoglobin was 41g/L. MK remained in ASCU for 2 days and returned to the ward, at which point her haemoglobin was 84g/L and further optimized with an iron infusion.
MK had been making a good post-operative recovery and by day 6 was considered suitable for discharge; however given the extent of her loss, the team opted for an additional day stay. On day 7, MK, complained of generalized abdominal pain and nausea. This was attributed to a lack of bowel motion for 6 days and she was given aperients with minimal relief of her nausea. MK remained haemodynamically stable, with a soft abdomen. Abdominal and pelvic ultrasounds were arranged to investigate the nausea and discomfort, as there was no clinical evidence of a collection or wound infection. Laboratory investigation revealed a profound hyponatremia with sodium of 114 mmol/L that was initially believed to be erroneous, but confirmed on repeat testing. Her haemoglobin was unremarkable at 105g/L. At that point midwifery staff informed the medical team of her confusion and that she had wandering around the ultrasound department earlier that day. It was also noted that although MK had managed to express colostrum, she had struggled to establish lactation.
Due to the profound hyponatremia, the medical team (obstetric physician and medical registrar) was contacted for an urgent consult. Collaboratively, the diagnosis of Sheehan’s syndrome was raised. MK was transferred to the intensive care unit of our affiliated tertiary hospital once stabilized. Her sodium level was slowly and carefully corrected with 3% saline and intravenous hydrocortisone replacement of 50mg fours times daily. MK returned back to the postnatal ward within two days. The remainder of her recovery was uncomplicated.
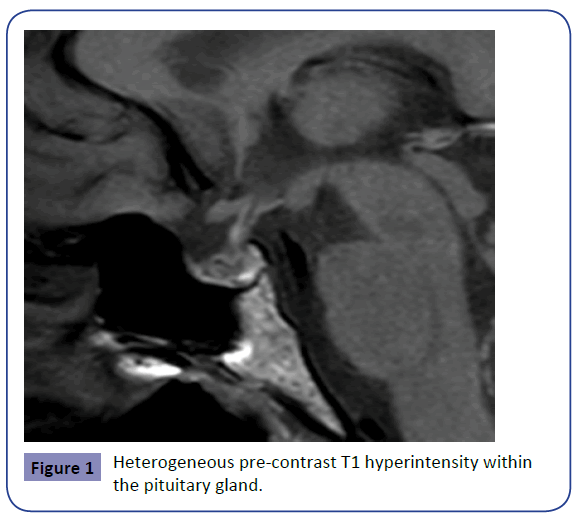
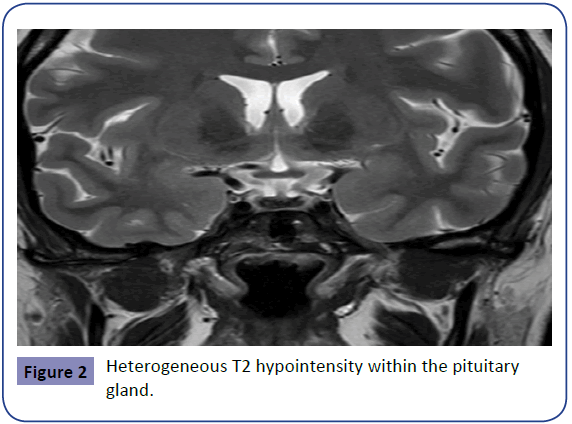
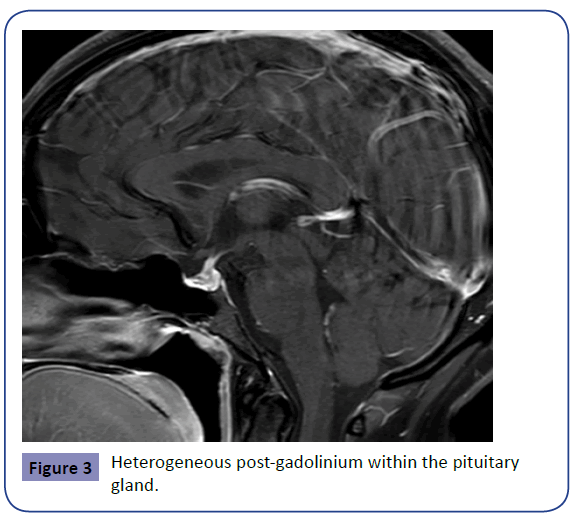
The initial MRI on day 8 demonstrated pre-contrast heterogeneous T1 hyperintensity within the pituitary gland and heterogeneous T2 central hypo-enhancing focus likely representing hemorrhage, however follow up was recommended to exclude underlying macroadenoma. The images below (1-3) demonstrate the findings of MK’s initial MRI (Figures 1-3).
The resolution of these findings on repeat MRI 6 months post-partum confirmed the diagnosis of haemorrhage/infarct (Sheehan’s syndrome) as opposed to adenoma, which would not have resolved. MK has continued the same dose of oral cortisone replacement commenced in hospital of 25mg twice daily. Although MK currently does not require thyroxine or oestrogen replacement her pituitary axis is being monitored by her endocrinology team, as pituitary function loss can be variable and develop over time, necessitating additional replacement. The table below documents MK’s pituitary axis 6 weeks post-partum. (Table 2).
| Hormonal Parameters | Patient value | Normal value |
|---|---|---|
| Serum T4 (mg/dl) Serum TSH (mIU/L) Cortisol (mg/dl) FSH (IU/L) LH (IU/L) E2 (pmol/L) Prolactin (mU/L) |
13 1.1 150 4 4.6 100 200 |
5.5-13.5 0.50-4.00 >18 Follicular 1-6. Post menopausal >20. Follicular 1-10. Post menopausal >20 Follicular 70-530. Post menopausal 0-120. <420 |
Table 2: Hormonal Parameters
Discussion
Why did MK develop hyponatraemia?
The clinical spectrum of presentation from Sheehan’s syndrome can vary considerably depending on the pituitary hormones affected by the infarction. In MK’s case, ACTH and consequent cortisol deficiency (secondary adrenal insufficiency) was of most clinical significance.
Acute cortisol deficiency can result in hyponatraemia through hypersecretion of ADH. There are two main mechanisms considered to contribute to over secretion of ADH in the setting of low cortisol; reductions in blood pressure and cardiac output in acute cortisol deficiency lead to ADH secretion and more importantly, corticotrophin releasing hormone (CRH) from the hypothalamus increasing in response to loss of cortisol. CRH acts as an ADH secretagogue [8].
The profound degree of hyponatraemia in this case is unusual in secondary adrenal insufficiency, however reflects the acute ischaemic sudden nature of the drop in ACTH. In the setting of acute cortisol loss, ADH is released and hyponatraemia develops (quite rapidly and profoundly). Once corrected with cortisol replacement, the inappropriate ADH release resolves and hyponatremia corrects [9].
The hyponatraemia was unlikely to be the result of aldosterone deficiency (as would be expected in primary adrenal deficiency), as MK was normovolaemic and normokalaemic during this period.
Although hypothyroidism chronically can cause hyponatremia, it rarely does so in an acute setting. MK’s thyroid function was normal during the admission and has remained so since, although the full pituitary deficit of Sheehan’s can develop over longer periods (months to years) [4-6].
How could this case impact future management?
Due to the rare occurrence of Sheehan’s syndrome in our population, there are no protocols for the ongoing surveillance and management of possible hypopituitarism. It remains important to consider Sheehan’s Syndrome in any patient who has endured a severe to massive PPH accompanied by periods of hypotension for assessment of adrenal insufficiency early in the post-partum period, as well as at 4-6 weeks post-partum [3] for any other hormonal deficiencies. If there are concerns, a multidisciplinary approach (Physician or Endocrinology) input is essential.
Acknowledgement
We would like to thank the midwifery and medical staff at King Edward Memorial Hospital for their work and ongoing commitment to their patients, as demonstrated in this case.
Conflict of Interest
The authors declare that there is no conflict of interest regarding the publication of this manuscript.
References
- Snyder PJ (2014) Causes of Hypopituitarism. In: UpToDate, Post, TW (1stedn), UpToDate, Waltham, MA.
- Hall E, Guyton AC (2011) Guyton and Hall textbook of medical physiology.
- Snyder PJ (2014) Clinical Manifestations of Hypopituitarism. In: UpToDate, Post, TW (1stedn), UpToDate, Waltham, MA.
- DökmetaÃÆââ¬Â¦Ãâà ¸ HS, Kilicli F, Korkmaz S, Yonem O (2006) Characteristic features of 20 patients with Sheehan's syndrome. Gynecol Endocrinol 22:279.
- Sert M, Tetiker T, Kirim S, Kocak M (2003) Clinical Report of 28 patients with Sheehan’s syndrome. Endocrine Journal 50: 297-301.
- Huang YY, Ting MK, Hsu BR, Tsai JS (2000) Demonstration of reserved anterior pituitary function among patients with amenorrhea after postpartum haemorrhage. Gynaecol Endocrinology 14: 99-104.
- Sanyal D, Raychaudhuri M (2012) Varied presentations of Sheehan’s syndrome at diagnosis: A Review of 18 patients. Indian J Endocrinol Metab16: S300-S301.
- Schrier RW (2006) Body water homeostasis: clinical disorders of urinary dilution and concentration. J Am Soc Nephrol 17:1820.
- Oelkers W (1989) Hyponatremia and inappropriate secretion of vasopressin (antidiuretic hormone) in patients with hypopituitarism. N Engl J Med 321: 492.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences